IONISING RADIATION is just one type of radiation. In physics, radiation describes any process in which energy travels through a medium or through, space, ultimately to be absorbed by another body.
In discussing the nuclear industry, we are not talking about other forms of radiation, which are mainly non-ionising – e.g: acoustic radiation (sound, ultrasound, infrasound) non-ionising forms of electromagnetic radiation (i.e., radio waves.)
While there is debate about whether or not electrmagnetic radiation is harmful to life, there is conclusive proof that ionising radiation is harmful.
RADIOACTIVE ISOTOPES: How they spread from the nuclear industry
The many stages of the nuclear cycle – at each stage, radiation is emitted. Transport of nuclear materials goes on between each stage.

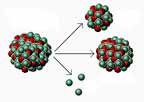
What is IONISING RADIATION?
Uranium atoms are unstable, breaking up into other atoms – radioisotopes, and giving off energy in the form of gamma rays
The 3 types of radiation (alpha, beta and gamma) are different in the way that they can travel through substances . (Alpha rays do not penetrate the body, but can be breathed in, or swallowed, and remain in tge body.) The radioactive isotopes also last for different lengths of time (see table below left)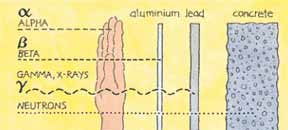
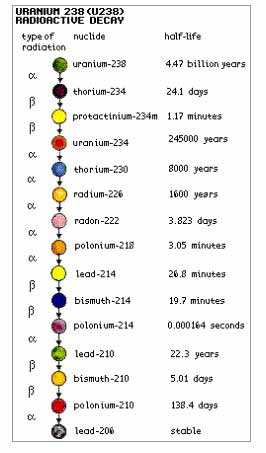 Radioactive isotopes vary in the time they take to break down.This length of time is measured in “half-lives” – counting the time it takes for an isotope to lose half of its radioactivity.
Radioactive isotopes vary in the time they take to break down.This length of time is measured in “half-lives” – counting the time it takes for an isotope to lose half of its radioactivity.
Some of the many toxic isotopes that are regularly released from nuclear reactors into water and air are Radioactive iodine 131, (half-life of 8 days), & Strontium 90 (half-life 28 days), Cesium 137 (half-life 30 years) is gradually released from ageing reactors.
Plutonium 239, created in nuclear reactors, is an alpha particle with a half-life of 24.400 years
Read more:
http://nuclear-news.net/information/wastes-2/
In discussing the nuclear industry, we are not talking about other forms of radiation, which are mainly non-ionising – e.g: acoustic radiation (sound, ultrasound, infrasound) non-ionising forms of electromagnetic radiation (i.e., radio waves.)
While there is debate about whether or not electrmagnetic radiation is harmful to life, there is conclusive proof that ionising radiation is harmful.
RADIOACTIVE ISOTOPES: How they spread from the nuclear industry
The many stages of the nuclear cycle – at each stage, radiation is emitted. Transport of nuclear materials goes on between each stage.


What is IONISING RADIATION?
Uranium atoms are unstable, breaking up into other atoms – radioisotopes, and giving off energy in the form of gamma rays
The 3 types of radiation (alpha, beta and gamma) are different in the way that they can travel through substances . (Alpha rays do not penetrate the body, but can be breathed in, or swallowed, and remain in tge body.) The radioactive isotopes also last for different lengths of time (see table below left)

 Radioactive isotopes vary in the time they take to break down.This length of time is measured in “half-lives” – counting the time it takes for an isotope to lose half of its radioactivity.
Radioactive isotopes vary in the time they take to break down.This length of time is measured in “half-lives” – counting the time it takes for an isotope to lose half of its radioactivity.Some of the many toxic isotopes that are regularly released from nuclear reactors into water and air are Radioactive iodine 131, (half-life of 8 days), & Strontium 90 (half-life 28 days), Cesium 137 (half-life 30 years) is gradually released from ageing reactors.
Plutonium 239, created in nuclear reactors, is an alpha particle with a half-life of 24.400 years
Read more:
http://nuclear-news.net/information/wastes-2/



